
Atomic valence electrons (shown in boxes on the left and right) fill the lower-energy molecular orbitals before the higher ones, just as is the case for atomic orbitals. At very long distances, there is essentially no difference in energy between the in-phase and out-of-phase combinations of H 1s orbitals. Demonstrate how Hü ckels theory approximates the full molecular orbital picture of molecules by treating the (sigma)-bonding and (pi)-bonding networks independently. This scheme of bonding and antibonding orbitals is usually depicted by a molecular orbital diagram such as the one shown here for the dihydrogen ion H 2 +. 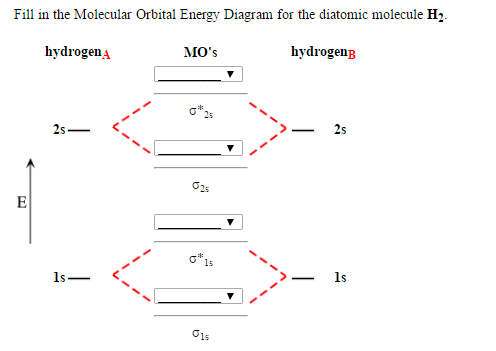
The hybridization of two atomic orbitals results in the formation of molecular orbitals. According to MOT number of atomic orbitals combined is equal to total. Use molecular orbital theory to predict the net bond order after N2 interacts with a photon that results in the promotion of an electron from bonding to an anti-bonding orbital. Remember that an orbital is a mathematical function that describes the probability of finding an electron in space. In order to predict the bond order, molecular orbital diagram for H2- is to be drawn. In this type of diagram, the molecular orbitals are represented by horizontal lines the higher a line the higher the energy of the orbital, and degenerate orbitals are placed on the same level with a space between them. Of course, there are 2s, 2p, 3s, 3p, etc. The energies of bonding and antibonding orbitals depend strongly on the distance between atoms. Hydrogen Molecular Orbital Diagram Atomic hydrogen has 1 electron in a 1s orbital. They will be unequal when there is an energy difference between the AOs, for example when a hydrogen 1s orbital and a chlorine 3p orbital combine to make a polar H-Cl bond. OF has 14 valence electrons, four in the 2p orbitals (see the diagram in the answer to Problem 5.9). The coefficients c 1 and c 2 will be equal (or nearly so) when the two AOs from which they are constructed are the same, e.g., when two hydrogen 1s orbitals combine to make bonding and antibonding MOs in H 2. The latter do not possess C2 rotation axes coincident to the infinite-fold rotation axis of the orbitals on the basis of the change in wave function sign upon crossing the nodes on the bond axis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
